 
In short, now you have to find the formal charge on the oxygen atom (O) as well as hydrogen atom (H) present in the OH molecule.įor calculating the formal charge, you have to use the following formula įormal charge = Valence electrons – (Bonding electrons)/2 – Nonbonding electrons The stability of lewis structure can be checked by using a concept of formal charge. Now you have come to the final step in which you have to check the stability of lewis structure of OH. Step 6: Check the stability of lewis structure Total valence electrons in OH- ion = valence electrons given by 1 oxygen atom + valence electrons given by 1 hydrogen atom + 1 more electron is added due to 1 negative charge = 6 + 1 + 1 = 8. You can see that only 1 valence electron is present in the hydrogen atom as shown in the above image. Hence the valence electron present in hydrogen is 1. Hydrogen is group 1 element on the periodic table. → Valence electrons given by hydrogen atom: You can see the 6 valence electrons present in the oxygen atom as shown in the above image. Hence the valence electrons present in oxygen is 6. Oxygen is group 16 element on the periodic table.  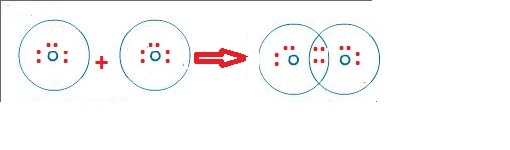
→ Valence electrons given by oxygen atom: Here, I’ll tell you how you can easily find the valence electrons of oxygen as well as hydrogen using a periodic table. (Valence electrons are the electrons that are present in the outermost orbit of any atom.) In order to find the total valence electrons in a OH- (hydroxide ion), first of all you should know the valence electrons present in a single oxygen atom as well as hydrogen atom. Steps of drawing OH- lewis structure Step 1: Find the total valence electrons in OH- ion 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
